From real-time patient data acquisition to intelligent interpretation, decision making and clinical action, integrated directly at the point of care, this marks the shift toward agentic AI in healthcare.
Everything in the Scanbo intelligence stack begins here — the hardware that captures clinical signals and the AI that interprets them. Deployed across 650+ settings. Generating revenue today.

19 lab-grade parameters at the bedside in under 5 minutes. No lab required. No qualified healthcare professional required. No temperature control required. Designed, manufactured, and deployed in-house with proprietary diagnostic strips.
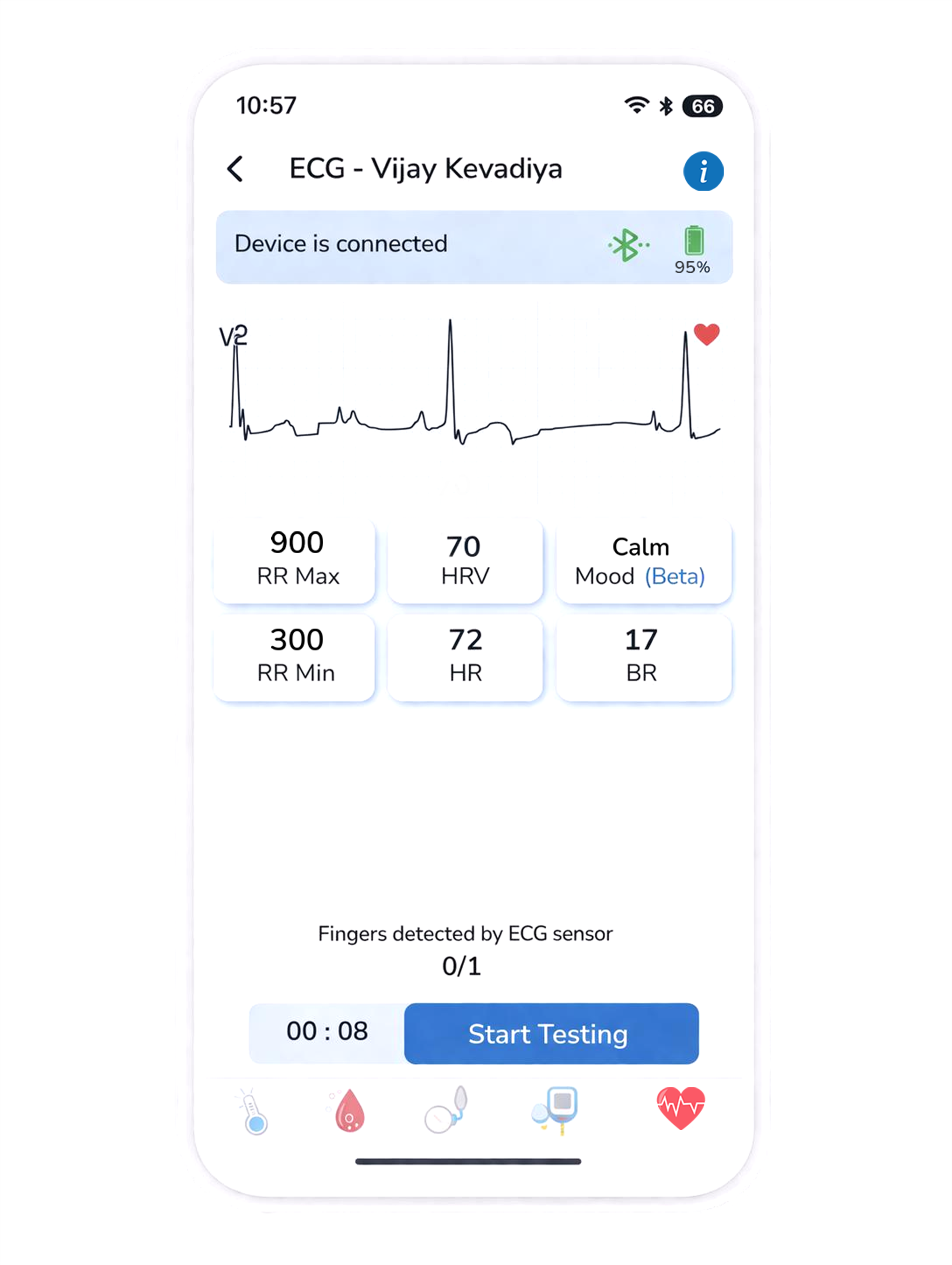
Single-lead ECG waveform interpretation powered by proprietary AI. Detects atrial fibrillation, sinus arrhythmia, missing heartbeats, and 8+ cardiac parameters in real-time. Deployed at scale. 1M+ analyses completed.
Each layer connects through FHIR R5 to the clinical data generated by D8 + HridayTaal.
Surfaces what mass conceals — visceral fat, skeletal muscle, and 40+ metabolic indicators captured directly into the FHIR R5 clinical record. 1M+ body scans recorded.
Autonomous conversion of clinical encounters into structured documentation. Zero transcription overhead. Eliminates documentation burden so clinicians focus on the patient.
TRY IT ↗Real-time drug interaction analysis and prescribing safety intelligence. Catches contraindications before they reach the patient. AI-powered medication safety at the point of prescribing.
TRY IT ↗The FHIR R5-native command center for clinical operations. Scheduling, telehealth, queuing, interoperability — all connected to the D8 signal layer and HridayTaal Cardiac Agent engine.
Our proprietary signal layer provides a clinical foundation for AI accuracy that cannot be replicated. Every metric below was generated directly through Scanbo hardware—in the room, at the moment of care.
Each D8 deployed is a data acquisition node feeding proprietary health signals into the intelligence stack. This signal layer cannot be purchased, licensed, or replicated — it can only be built by owning the hardware in the room, at the moment of care.
Every device in the field is a data acquisition node. The moat widens every month.
Not licensed, not aggregated, not scraped. Generated by Scanbo hardware at the point of care.
Signal volume, device count, and revenue all compounding year-on-year.
Device to EMR in seconds. Every reading captured, interpreted, and recorded at the point of consultation. No transcription. No delay. No errors.
Documentation time eliminated across 50 devices in a diabetes reversal network.
Point-of-consultation vitals with auto-push to EMR. Clinical note quality measurably improved.
Accountable Care Organisations in Florida and New York evaluated the full clinical stack — data security, integration, and outcomes — and chose to pilot before regulatory clearance. US commercialisation is already in motion.
Five peer-reviewed studies confirm Scanbo®'s diagnostic accuracy in real-world deployments — indexed on PubMed, ScienceDirect, and recognised by PATH and WHO-aligned global health programmes.
Confirms Scanbo® D8 measures blood pressure and glucose to clinical standards in a low-resource urban deployment — no lab, no specialist infrastructure required.
Peer-reviewed publication in a Lancet-family digital health journal — the highest-credibility venue for point-of-care diagnostic research. Examines outcomes across real healthcare environments.
Confirms the research is independently indexed, citable, and discoverable across global academic platforms — a signal of reproducible, peer-traceable clinical science.
PATH — a WHO-aligned global health organisation — selected Scanbo® as a featured technology in its evaluation of AI-powered diagnostic tools for next-generation primary healthcare delivery.
Authoritative PATH report on AI-powered diagnostic innovation in global primary care. A credible third-party reference for investors, health system partners, and procurement leads evaluating Scanbo®'s real-world context.
Scanbo® has earned coverage across major business, health, and technology media — from executive interviews and authored articles to multi-outlet national wires and international trade press.
Scanbo® generates revenue across hardware consumables, SaaS subscriptions, and proprietary AI — with all three streams active and growing year-on-year.
Diagnostic test-based SaaS pricing — clinics and hospitals pay per test volume, not per seat. Scales with usage.
Like a printer and its cartridges — every diagnostic test requires a proprietary strip, creating a recurring revenue stream tied to each device in the field.
Clinical AI Licensing — 30+ proprietary algorithms licensed to health systems as decision-support tools. The intelligence is the product. Patient data is never sold.
Last updated: March 2026
Scanbo® Technologies, Inc. ("Scanbo®", "we", "us", or "our") is a point-of-care health technology company headquartered in Canada with operations across India, the US, North America, and the EU. We develop medical devices, clinical AI software, and connected health platforms.
For EU users, we process data under: contract performance, legal obligation, legitimate interests, and explicit consent where required. You may withdraw consent at any time.
We do not sell your personal information. We share data only with trusted third-party service providers under strict data processing agreements, or as required by law.
We retain personal data only as long as necessary for the purposes described or as required by applicable law, including medical records regulations in each jurisdiction.
Depending on your jurisdiction, you may have the right to: access, correct, delete, or port your data; object to or restrict processing; withdraw consent; and file a complaint with a supervisory authority.
We implement industry-standard technical and organisational measures including encryption at rest and in transit, access controls, and regular security audits.
Last updated: March 2026
By accessing or using any Scanbo® product, platform, website, or service, you agree to be bound by these Terms. If you do not agree, please discontinue use immediately.
Scanbo® products include medical devices (Scanbo® D8, Smart Scale), clinical AI software (HridayTaal® Cardiac Agent, RxShield Safety Agent, Ambient Documentation Agent), and connected platforms (Scanbo® EMR). These are intended for licensed healthcare professionals and organisations only, unless explicitly stated otherwise.
Scanbo® products are decision-support tools. They are not intended to replace the professional judgment of a licensed clinician. All clinical decisions must be made by a qualified healthcare professional.
All Scanbo® trademarks, patents, software, algorithms, AI models, hardware designs, and content are the exclusive intellectual property of Scanbo® Technologies, Inc. Unauthorised reproduction or reverse engineering is strictly prohibited.
To the maximum extent permitted by law, Scanbo® shall not be liable for any indirect, incidental, special, or consequential damages arising from use or inability to use our products or services.
These terms are governed by the laws of British Columbia and the applicable federal laws of Canada. Disputes shall be resolved in British Columbia, Canada, unless a mandatory local law requires otherwise. For EU customers, applicable EU consumer protection laws apply.
Last updated: March 2026
Cookies are small text files placed on your device when you visit our website. They help us operate the site, understand how it is used, and personalise your experience.
Required for the website to function. Cannot be disabled. Includes session management and security tokens.
Help us understand how visitors interact with our website. Data is aggregated and anonymised.
Remember your preferences such as language, region, and login state to improve your experience.
Used to serve relevant content and measure campaign effectiveness. Requires explicit consent.
You can update cookie preferences at any time via your browser settings or by contacting us. Disabling certain cookies may affect site functionality.
Last updated: March 2026
Scanbo® is committed to providing equal access to all individuals regardless of race, colour, national origin, age, disability, sex, gender identity, sexual orientation, religion, or any other characteristic protected under applicable law.
Our clinical AI algorithms are trained and validated across diverse populations to minimise bias. Ambient Documentation Agent supports 11 languages to reduce language barriers. Our devices are designed for both urban and rural settings.
Scanbo® provides equal employment opportunities to all. We do not discriminate in hiring, compensation, promotion, or termination based on any protected characteristic.
We strive to make digital products accessible in compliance with WCAG 2.1 AA. If you experience accessibility barriers, please contact us.
Last updated: March 2026
We welcome reports from security researchers and clinicians about potential vulnerabilities in our products and systems. We take security of medical devices and patient data extremely seriously.
Please report before public disclosure to allow us to investigate and remediate. We will acknowledge within 5 business days, investigate and keep you informed, and will not pursue legal action against good-faith researchers.
Applies to: Scanbo® D8 · Smart Scale · HridayTaal® Cardiac Agent · Last updated: March 2026
Scanbo® diagnostic products are intended for use by licensed healthcare professionals in clinical settings. They are not intended for home use by untrained individuals or as a replacement for laboratory diagnostics unless explicitly stated.
Store at 10–40°C, 15–85% relative humidity, non-condensing. Full maintenance schedules are in the device-specific User Manual shipped with each product.
Effective: March 2026 · Applies to US customers
Where Scanbo® acts as a Business Associate to US-based Covered Entities, we comply with HIPAA 1996 and its implementing regulations including the Privacy Rule, Security Rule, and Breach Notification Rule.
As a Business Associate, Scanbo® uses Protected Health Information only to provide contracted services to the Covered Entity, as required by our BAA, or as required by law.
US healthcare organisations must execute a BAA prior to go-live. Contact us to request a BAA.
In the event of a PHI breach, Scanbo® will notify the affected Covered Entity within 60 calendar days of discovery per the HITECH Act.
Effective: March 2026 · Applies to EEA/UK customers
Scanbo® is committed to full compliance with the General Data Protection Regulation (EU) 2016/679 and the UK GDPR for all personal data of individuals in the EEA and United Kingdom.
Transfers from the EEA to India use Standard Contractual Clauses approved by the European Commission.
You may lodge a complaint with your local supervisory authority. Full list at edpb.europa.eu.
Digital Personal Data Protection Act, 2023 · Last updated: March 2026
Scanbo® is committed to compliance with India's DPDP Act, 2023. As a company headquartered in India processing personal data of Indian citizens, this Act applies directly to our operations.
Under the DPDP Act, Scanbo® is a Data Fiduciary — we determine the purpose and means of processing personal data. For clinical data processed on behalf of healthcare providers, we may also act as a Data Processor.
Last updated: March 2026
Under GDPR, HIPAA, and India's DPDP Act, you have the right to access, correct, delete, or receive a copy of your personal data held by Scanbo®.
Receive a copy of all personal data we hold about you.
Update or correct inaccurate personal data we hold.
Request erasure of your personal data subject to legal obligations.
Receive your data in a structured, machine-readable format.
All pages across the Scanbo® platform
Guidelines for using RxShield Safety Agent in telehealth and virtual care settings across the US and Canada. Last updated: March 1, 2026
RxShield Safety Agent is a reference tool, not a telehealth platform. It does not facilitate patient communication, video consultations, electronic prescribing, or any patient-facing service.
This policy applies to licensed healthcare professionals using RxShield Safety Agent in connection with telehealth or virtual care services in the US and Canada.
In emergency scenarios, always prioritize direct patient assessment and established emergency protocols. RxShield Safety Agent is not designed for emergency decision support.
Telehealth prescribing is governed by state-specific laws and the Ryan Haight Act for controlled substances. Clinicians must comply with licensing and prescribing obligations in the state where the patient is located.
Virtual care prescribing is governed by the clinician's provincial licensing body. Clinicians must comply with provincial College guidelines on virtual prescribing.
The standard of care applicable to a telehealth encounter is the same as, or may be higher than, an in-person visit. The use of RxShield Safety Agent does not modify this standard.
Clinicians using RxShield Safety Agent in telehealth encounters are responsible for documenting their prescribing decisions in the patient record. RxShield Safety Agent Decision IDs may be used as supplementary documentation of evidence reviewed.
Where RxShield Safety Agent is integrated with an EMR in a telehealth environment, the clinician remains responsible for reviewing and confirming any data passed between systems.
For questions about this policy, contact us at legal@scanbo.com
In no event shall Scanbo™ be liable for any damages (including, without limitation, lost profits, business interruption, or lost information) arising out of your use of or inability to use the product, even if Scanbo™ has been advised of the possibility of such damages. No distributor, dealer or other party is authorized to make any warranty on behalf of Scanbo™, or to assume for Scanbo™ any other liability with respect to its products, services, data storage and protection.
Scanbo™ is not liable for any inaccurate, mistaken or false results that are shown on the products. Scanbo™ is not an alternative to any medical procedure or check-up that would require qualified doctors. To the extent that the product and the information and services are provided free of charge, we will not be liable for any loss or damage of any nature.
We will not be liable to you in respect of any business losses, including (without limitation) loss of or damage to profits, income, revenue, use, production, anticipated savings, business, contracts, commercial opportunities or goodwill. We will not be liable to you in respect of any loss or corruption of any data, database or software. We will not be liable to you in respect of any special, indirect or consequential loss or damage.
Except for the express warranty set forth herein, the product and software provided hereunder are provided "as is" and Scanbo™ makes no warranty as to its use or performance. Except for any warranty, condition, representation or term the extent to which cannot be excluded or limited by applicable law, Scanbo™ and its suppliers make no warranty, condition, representation, or term (express or implied, whether by statute, common law, custom, usage or otherwise) as to any matter including, without limitation, non-infringement of third party rights, merchantability, integration, or fitness for a particular purpose.
You assume responsibility for selecting the product to achieve your intended results, and for the installation of, use of, and results obtained from the product and software. Without limiting the foregoing provisions, Scanbo™ makes no warranty that the equipment or software will be error-free or free from interruptions or other failures or that the software or product will meet your requirements.
Under no circumstances and under no legal theory, whether in tort, contract, or otherwise, shall Scanbo™ or its suppliers be liable to the purchaser or to any other person for damages for gross negligence of any character including, without limitation, damages for loss of goodwill, work stoppage, computer failure or malfunction, or for any other damage or loss. In no event shall Scanbo™ or its suppliers be liable for any damages in excess of the price paid for the product and software, even if Scanbo™, or its authorized partners or suppliers have been advised of the possibility of such damages.
User grants us the right to use, copy, store, transmit and display User data solely to the extent necessary to provide the Platform and associated maintenance, for purposes of ensuring security, and for the investigation of complaints. All data submitted by User to the Platform (User Data) will remain the sole property of User. Subject to the terms of this Agreement, we shall not disclose User Data or use User data for any purpose other than to provide the Platform and as set forth herein.
The Platform uses "cookies", which are small text files placed on the User's hard disk by a Web server. Cookies contain information that can later be read by a Web server in the domain that issued the cookie to the User. One of the primary purposes of cookies is to store the User's preferences and other information on the User's computer in order to save the User's time by eliminating the need to repeatedly enter the same information and to display the User's personalized content and targeted advertising on the User's later visits to this Platform.
When the User signs in the Platform named User account, we store the User's unique ID number, and the time the User signed in, in an encrypted cookie on the User's hard disk. This cookie allows the User to move from page to page at the Platform without having to sign in again on each page. When the User signs out, these cookies are deleted from the User's computer. We also use cookies to improve the sign-in experience. User agrees that we may use Usage Statistics to provide Users with communications regarding Platform support, news and information regarding the Platform and services/products provided by us, except where such Users opt out of receiving such information.
For questions about this disclaimer, contact us at legal@scanbo.com
A small focused team building clinical devices, AI, and software used in real healthcare settings. If you want your work to matter, we are hiring.
We review every application at hr@scanbo.com
Thank you — we will be in touch at hr@scanbo.com
Whether you are a clinician, health system, investor, or partner — we would like to hear from you.
We respond to every message — typically within 1–2 business days.
Thank you for reaching out. Our team will follow up at your email address within 1–2 business days.
How real organizations turn point-of-care intelligence into clinical and operational outcomes.
Scanbo® D8 captures eight vital parameters in minutes and pushes data directly into the EMR. Supraja eliminated manual transcription across OPD and IPD — the time recovered goes back to patient care.
WWC Clinics deployed Scanbo® D8, Smart Scale, and EHR across their chronic care network. Video consultation and secure messaging extended clinical reach significantly — patients receive continuous monitoring without returning to the clinic for every check-in.
In a high-volume neurology outpatient setting, Scanbo® D8 delivers reliable multiparameter vitals at the moment of consultation. Every reading is captured and recorded instantly into the EMR — no transcription lag, no manual errors. Clinical note quality measurably improved.
Measurably reduced documentation time across OPD and IPD. That time goes back to patient care.
The EMR integration means every reading is recorded instantly — no manual entry, no errors. It has genuinely improved the quality of my clinical notes.
D8, Smart Scale, and EMR work as one system. Patients get continuous monitoring without needing to visit every time.
We work with health systems, chronic care providers, diagnostic programmes, and community health organisations.
The deployed foundation of Vitals Intelligence. One connected device capturing 8 clinical parameters at the point of care — no lab, no manual entry, no specialist infrastructure. Already running across 650+ clinical settings as the first layer of the Scanbo® platform.



Most point-of-care tools are single-parameter or lab-bound. Scanbo® D8 is different: one connected device that captures, interprets, connects, and protects clinical data — all in a single workflow.
Where most devices capture one reading, D8 gives a clinician a full picture in a single session — blood pressure, ECG, SpO2, glucose, and more. Faster decisions. Fewer gaps.
D8 feeds directly into Scanbo® EMR. ECG data routes to HridayTaal® Cardiac Agent for interpretation. Every reading becomes a data point in the care continuum — no manual entry, no transcription errors.
CDSCO approved and validated in real urban field settings. Purpose-built for pharmacies, clinics, community health workers, and frontline care — not just hospital wards. Already deployed across India at scale.
Every parameter D8 captures is billable, documentable, and immediately available in the clinical record. No lab turnaround. No separate device. No manual entry.
Each test uses a proprietary Scanbo® strip — ensuring consistent, calibrated results for every patient assessment.
The D-Series is Vitals Intelligence expanding over time. Each generation is built on the same architecture, workflows, and AI infrastructure. Deploying D8 today means being platform-ready for D12, D16, and D19 — without switching systems.
D8 is built for the settings conventional tools struggle to serve — high-volume OPDs, distributed community care, and value-based programs that need real clinical data at the point of assessment.
Reduces documentation time, eliminates manual entry errors, and feeds data directly into Scanbo® EMR. Already deployed in neurology, diabetes, and general OPD settings.
D8 qualifies under existing Medicare SMBP and RPM billing frameworks. No new CPT code required. Strong fit for ACOs and value-based care programs deploying remote monitoring.
Validated in low-resource urban deployments. No lab, no specialist infrastructure. Community health workers screen patients and capture data directly into the clinical record.
D8 qualifies across SMBP, RPM, ECG, and physician review billing pathways. CMS already recognises every parameter D8 captures. Full code details are in the Reimbursement Guide.
Independent research confirms Scanbo® D8 diagnostic accuracy in actual clinical environments — not controlled lab conditions. Published across PubMed, PMC, ScienceDirect, and Semantic Scholar.
Book a live demo tailored to your organisation — clinics, ACOs, health systems, and community health programmes welcome.
Book a demo →Scheduling, communication, and interoperability in one connected platform. Scanbo® EMR is not just a records system — it is the workflow layer that helps care actually run, with FHIR R5-native interoperability built in.
Most EHR systems are built around documentation. Scanbo® EMR is built around workflow — the scheduling, communication, and operational visibility that keeps care running smoothly, connected to every other layer of the Scanbo® platform.
Booking, rescheduling, cancellation, and waitlisting — all within the same platform. No handoffs to external scheduling tools. No manual coordination between systems.
A live queue dashboard gives clinical and administrative staff visibility into who is waiting, who is in consultation, and what is delayed — reducing friction and improving patient throughput.
Scanbo® D8 vitals, HridayTaal® ECG interpretations, and Smart Scale body composition all flow directly into the EHR. Every clinical encounter is connected, documented, and longitudinally visible.
From first booking to live queue status, Scanbo® EMR gives clinical teams the scheduling infrastructure and operational visibility to run high-volume care settings without operational drag.
Clinical communication should not live in a separate app. Scanbo® EMR includes HIPAA-compliant audio TeleCall, video TeleCall, and Secure Chat built into the same workflow environment.
Browser-native audio calling via WebRTC. No app download required. Patients connect from any device — audio quality is stable, and the call is logged against the patient record.
Full video consultation within the EHR environment. Clinicians can view vitals from D8, review ECG interpretations, and conduct the consultation — all in the same screen without switching tools.
Encrypted messaging between clinicians, care teams, and patients. Secure Chat replaces insecure workarounds like WhatsApp or SMS for clinical communication, with messages logged in the patient record.
Scanbo® EMR is built with FHIR R5-native interoperability — not added on, not planned for later. For organizations still operating on older FHIR versions, built-in migration and conversion tooling provides a clear, manageable path toward FHIR R5 without unnecessary disruption.
D8 Vitals Intelligence, HridayTaal® Cardiac Intelligence, and Smart Scale Metabolic Intelligence all feed directly into Scanbo® EMR. Every reading, every interpretation, every measurement — connected, documented, and longitudinally visible in one place.
Book a live demo to see how Scanbo® EMR connects scheduling, communication, and FHIR R5 interoperability in one platform.
Book a demo →Scanbo® manufactures its own devices under direct quality control, with ISO 13485-aligned systems, MDSAP audit readiness, and an active FDA 510(k) programme — the operational foundation that makes consistent, scalable deployment possible.
In regulated healthcare environments, manufacturing and quality control are foundational — not secondary. Consistency, traceability, and audit-readiness are what make it possible to deploy at scale, enter new markets, and earn the trust of clinical and enterprise partners.
Scanbo® manufactures its own devices in-house, with full control over production quality, component sourcing, traceability, and delivery — no external contract manufacturers.
Quality management systems cover every stage from incoming components to final device release. Each unit is traceable, each process documented, each deviation controlled.
Scanbo®’s regulatory programme supports market access in India, Canada, and the United States — aligned to CDSCO approval, Health Canada review, and FDA 510(k) submission.
The international quality management standard for medical devices. Scanbo®’s operations are aligned to ISO 13485 across design, production, and post-market processes.
A single audit programme covering regulatory requirements across Canada, the United States, Brazil, Australia, and Japan.
Scanbo® D8 is on track for FDA 510(k) clearance in Q1 2027. The quality infrastructure built toward this submission is the same system that governs every Scanbo® device — not a separate process created for regulatory review.
For healthcare providers, enterprise buyers, and institutional partners, Scanbo®’s manufacturing and quality infrastructure is the operational assurance behind every device and deployment. In-house production, ISO 13485-aligned systems, and an active regulatory programme are the foundation that makes consistent, scalable deployment possible.
Scanbo® is a connected point-of-care intelligence platform. We build the infrastructure that closes the gap between a health signal and a clinical decision — at the point of care, not after it.
Across thousands of OPDs, community clinics, and distributed care programmes, the same failure repeats: health data is captured but not connected, measured but not interpreted, recorded but not acted on. The tools do not talk to each other. The patient pays the cost.
Vitals Intelligence. Cardiac Intelligence. Metabolic Intelligence. Workflow Intelligence. Each layer captures, interprets, and connects. Together they form a clinical infrastructure that gets smarter with every deployment, every patient, every day.
This is not a hypothesis. It is a deployed, revenue-generating platform with clinical evidence and commercial traction.
From real-time patient data acquisition to intelligent interpretation, decision making and clinical action, integrated directly at the point of care, this marks the shift toward agentic AI in healthcare.
A world where no patient loses time because the insight that could have helped them was trapped in a silo instead of reaching the clinician.
FDA 510(k) clearance is on track for Q1 2027. Health Canada review is underway. The same intelligence architecture running across 650+ clinical settings in India is being brought to ACOs, health systems, and value-based care programmes in North America — without rebuilding from scratch.
Scanbo® D8 and Smart Scale qualify under existing Medicare SMBP and RPM billing frameworks. Deploy now using established pathways — no new code required.
No new CPT code required. CMS already recognises weight, blood pressure, pulse oximetry, and respiratory flow rate as billable RPM parameters — the exact parameters Scanbo® D8 captures. Providers deploy Scanbo® inside billing frameworks they already use.
Built for reimbursable SMBP, RPM, and rhythm-monitoring workflows. The broadest reimbursement story of the two products.
Clean RPM-first entry point. CMS materials specifically cite weight as a remote physiologic monitoring parameter — perfect positioning.
National average non-facility rates · Source: CMS Physician Fee Schedule · Effective January 1, 2026
Illustrative revenue potential per year, based on 2026 CMS national average non-facility rates.
Key billing rule: RPM codes bill once per patient per 30-day period — regardless of device count. D8 + Smart Scale on the same patient = one RPM billing set. Figures below model separate patient cohorts per product.
Scanbo® is a deployed, revenue-generating point-of-care intelligence platform expanding into North America. We hold structured briefings with qualified investors and strategic institutional partners.
We hold structured briefings with qualified investors and strategic partners. Complete the form and our leadership team will follow up directly.
All requests are reviewed by our leadership team. Your information is kept strictly confidential.
Our leadership team will review your request and follow up within 2–3 business days to schedule a call.
The Metabolic Intelligence layer. Every measurement generates an AI Body Score — a single, trackable signal informed by visceral fat, skeletal muscle, subcutaneous fat, and metabolic indicators — building longitudinal insight directly into the Scanbo® platform.
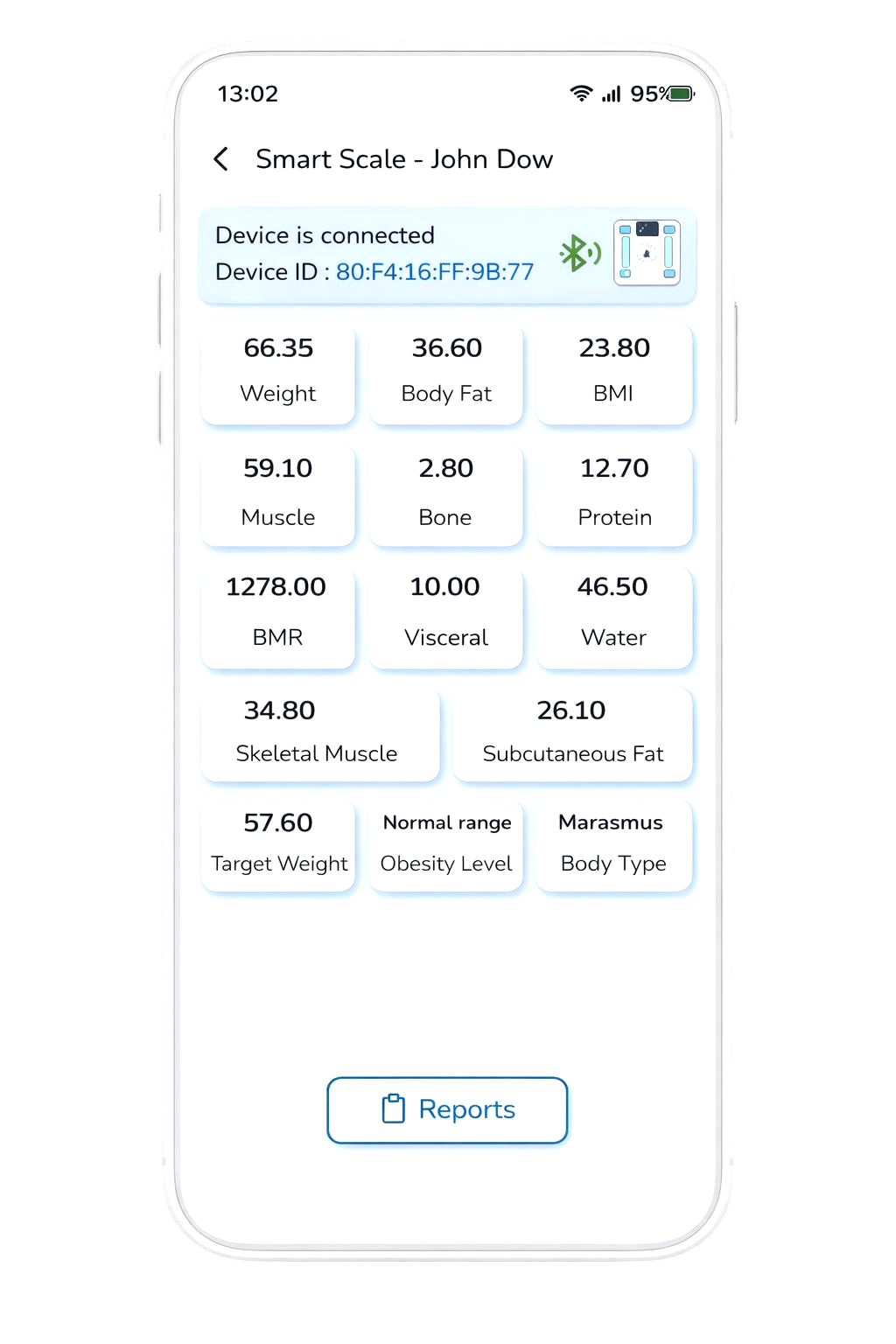

Consumer scales measure weight. Scanbo® Smart Scale surfaces what weight conceals — visceral fat, subcutaneous fat, skeletal muscle, body water, and an AI-driven body score — connecting metabolic insight directly into the clinical record.
Each measurement generates an AI Body Score — a composite signal informed by visceral fat, subcutaneous fat, skeletal muscle, body water, protein, and bone mineral mass. One number that summarises metabolic state.
A single reading tells you a number. Longitudinal tracking tells you a direction. Body Score trends over time reveal whether metabolic health is improving, stable, or declining — informing clinical decisions before conditions worsen.
Every reading flows automatically into Scanbo® EMR — available to the clinical team at every touchpoint, without manual entry or patient recall.
Smart Scale is not a wellness gadget. It is the Metabolic Intelligence layer inside the Scanbo® platform — sitting alongside D8 Vitals Intelligence, HridayTaal® Cardiac Intelligence, and Scanbo® EMR — giving clinicians visceral fat trends, body score changes, and metabolic shifts between visits.
Use D8 to capture cardiovascular and metabolic vitals. Use Smart Scale to capture body composition. Together they give the clinician a more complete picture at the point of care.
Patients measure at home. Data flows automatically into the clinical record. Clinicians see composition trends between visits — enabling earlier intervention and more informed consultations.
Weight monitoring qualifies under existing CMS RPM billing pathways. No new code required. Smart Scale fits directly into the same reimbursement workflow as D8.
Already deployed across diabetes reversal and chronic disease management clinics. Body composition trends are clinically meaningful in metabolic care — Smart Scale makes them accessible.
CMS recognises body weight as a billable RPM parameter. Smart Scale deployments qualify under the same 99453, 99454, and 99457 pathways used by Scanbo® D8. Full details in the Reimbursement Guide.
Book a live demo to see how Smart Scale and D8 work together inside the Scanbo® platform.
Book a demo →The Analyst interprets raw ECG signals to identify arrhythmias.
The deployed Cardiac Intelligence layer. HridayTaal® turns a single-lead ECG into structured rhythm insight at the point of care — connected to the Scanbo® platform, and expanding its scope over time.
Standard point-of-care ECG tools display a trace. HridayTaal® goes further — applying AI to identify rhythm patterns, surface irregularities, and return structured output that supports clinical decision making.
HridayTaal® takes a raw single-lead ECG trace and returns clinically relevant rhythm interpretation — automatically, at the point of care.
No specialist required for initial rhythm assessment. HridayTaal® delivers AI interpretation at the front line — in clinics, pharmacies, and community care settings.
Interpretation outputs connect directly into Scanbo® EMR. Every rhythm insight is documented, timestamped, and longitudinally visible alongside the patient record.
These capabilities are live in clinical deployment, running on every ECG captured through Scanbo® D8.
A single-lead ECG at the point of care is only as useful as the interpretation layer behind it. HridayTaal® turns a raw trace into actionable structured output — without requiring a cardiologist at every location.
Clinicians receive structured rhythm output with each ECG, giving them the information needed to triage, escalate, or continue monitoring — without waiting for downstream specialist review.
Because every interpretation is recorded in Scanbo® EMR, rhythm trends emerge over multiple visits. Intermittent arrhythmias become visible. Progression becomes detectable.
In high-volume primary care settings, AFib detection at the point of consultation has significant population health value. HridayTaal® makes routine ECG screening clinically useful, not just technically possible.
Community health workers and primary care clinicians gain structured cardiac interpretation previously available only in specialist settings. This expands meaningful ECG use across all care levels.
Cardiac Intelligence is expanding over time. Each generation of HridayTaal® adds deeper signal analysis — extending the rhythm insight available at the point of care.
These capabilities are in active pre-clinical research and are not yet deployed or validated for clinical use.
Every ECG captured by Scanbo® D8 is automatically analysed by HridayTaal®. Rhythm interpretation flows into Scanbo® EMR alongside vitals, body composition, and longitudinal patient data — giving clinicians a connected picture, not isolated readings.
HridayTaal® is a decision-support layer. It provides structured rhythm output to assist clinically trained professionals — it does not replace physician judgement.
Running on every Scanbo® D8 ECG capture across 650+ clinical settings in India. Over 1M+ ECG analyses completed in active deployment.
Interpretation outputs are structured for clinical documentation and review. HridayTaal® is designed to augment, not automate, clinical cardiac assessment.
Book a live demo to see how HridayTaal® interprets ECG data in real time alongside Scanbo® D8 and the connected Scanbo® platform.
Book a demo →Our team will walk you through a live demo tailored to your organisation — hospitals, clinics, ACOs, or health systems welcome.
Our team typically responds within 2–3 business days. No spam, ever.
Thank you — we'll be in touch within 2–3 business days.
The URL you entered is not part of this website. Please check the path or return to the home page.
Requested route: /404